Azor 10 40mg12/30/2023 
Patients with hypovolaemia or sodium depletion: The tablet should not be chewed and should be taken at the same time each day. The tablet should be swallowed with a sufficient amount of fluid (e.g. The safety and efficacy of Sevikar in children and adolescents below 18 years has not been established. Use of Sevikar in patients with severe hepatic impairment is contraindicated (see section 4.3). Amlodipine should be initiated at the lowest dose and titrated slowly in patients with impaired liver function. The pharmacokinetics of amlodipine have not been studied in severe hepatic impairment. Sevikar should therefore be administered with caution in these patients. There is no experience of olmesartan medoxomil in patients with severe hepatic impairment.Īs with all calcium antagonists, amlodipine's half-life is prolonged in patients with impaired liver function and dosage recommendations have not been established. Close monitoring of blood pressure and renal function is advised in hepatically-impaired patients who are already receiving diuretics and/or other antihypertensive agents. In patients with moderate hepatic impairment, an initial dose of 10 mg olmesartan medoxomil once daily is recommended and the maximum dose should not exceed 20 mg once daily.  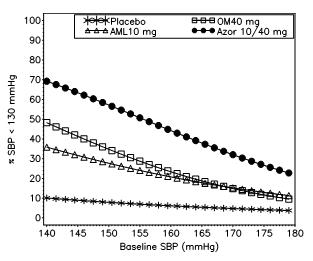
Sevikar should be used with caution in patients with mild to moderate hepatic impairment (see sections 4.4, 5.2). Monitoring of potassium levels and creatinine is advised in patients with moderate renal impairment. The use of Sevikar in patients with severe renal impairment (creatinine clearance < 20 mL/min) is not recommended (see 4.4, 5.2). The maximum dose of olmesartan medoxomil in patients with mild to moderate renal impairment (creatinine clearance of 20 – 60 mL/min) is 20 mg olmesartan medoxomil once daily, owing to limited experience of higher dosages in this patient group. If up-titration to the maximum dose of 40 mg olmesartan medoxomil daily is required, blood pressure should be closely monitored. No adjustment of the recommended dose is generally required for elderly people but increase of the dosage should take place with care (see sections 4.4 and 5.2). Sevikar can be taken with or without food. When clinically appropriate, direct change from monotherapy to the fixed combination may be considered.įor convenience, patients receiving olmesartan medoxomil and amlodipine from separate tablets may be switched to Sevikar tablets containing the same component doses. Sevikar 40 mg/10 mg may be administered in patients whose blood pressure is not adequately controlled by Sevikar 40 mg/5 mg.Ī step-wise titration of the dosage of the individual components is recommended before changing to the fixed combination. Sevikar 40 mg/5 mg may be administered in patients whose blood pressure is not adequately controlled by Sevikar 20 mg/5 mg. Sevikar 20 mg/5 mg may be administered in patients whose blood pressure is not adequately controlled by 20 mg olmesartan medoxomil or 5 mg amlodipine alone. The recommended dosage of Sevikar is 1 tablet per day. Date of first authorisation/renewal of the authorisation 6.6 Special precautions for disposal and other handling.4.7 Effects on ability to drive and use machines.4.5 Interaction with other medicinal products and other forms of interaction.4.4 Special warnings and precautions for use.4.2 Posology and method of administration.Medically administered drugs are prescription medications and vaccines that are typically administered by a health care provider in the medical office.Show table of contents Hide table of contents 
Non-Formulary Brand drugs are drugs that are not included on the plan’s list of preferred prescription drugs. These drugs are produced and sold under the original manufacturer’s name. If prescribed a brand-name drug, you’ll get a generic substitute unless there isn’t a generic substitute or Kaiser Permanente approves use of the brand-name drug.įormulary Brand drugs are drugs that are included on the benefit plan’s list of preferred prescription drugs. The quality and effectiveness of generic drugs is the same as brand name drugs. Generic drugs are determined by The Federal Food and Drug Administration (FDA) which requires that generic drugs contain the same active ingredients in the same amount as the brand name drug. Non-formulary brands drugs are covered at the Cateogry 3 coverage amount. Formulary brand drugs will be covered at the Cateogry 2 coverage amount. Generics drugs are those covered at the lowest cost share defined as Cateogry 1. A 3-tier system is used to determine your cost share.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
